- Methasterone
-
Methasterone 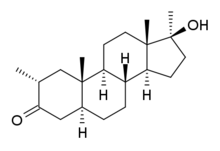
Systematic (IUPAC) name 17β-Hydroxy-2α,17α-dimethyl-5α-androstane-3-one Clinical data Pregnancy cat. ? Legal status ? Routes Oral Identifiers CAS number 3381-88-2 
ATC code None Synonyms Superdrol, methyldrostanolone Chemical data Formula C21H34O2 Mol. mass 318.492 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Methasterone (Superdrol, methasteron, and methyldrostanolone) is an oral anabolic steroid that was never marketed through legitimate channels for medicinal purposes. It was brought to market, instead, in a clandestine fashion as a “designer steroid.”
Contents
History
The synthesis of methasterone is first mentioned in the literature in 1956 in connection with research conducted by Syntex Corporation in order to discover a compound with anti-tumor properties.[1] This initial mention is elaborated upon in a 1959 research journal article, where its method of synthesis is discussed in greater detail, its tumor inhibiting properties are verified, and it is noted as being a “potent orally active anabolic agent exhibiting only weak androgenic activity.”[2] The results of subsequent assays to determine methasterone’s anabolic and androgenic activity were published in Vida’s Androgens and Anabolic Agents, a dated but still standard reference, where it was noted that methasterone possessed the oral bioavailability of methyl-testosterone while being 400% as anabolic and 20% as androgenic, yielding a Q-ratio (also known as an anabolic to androgenic ratio) of 20, which is considered very high.[3]
Injectable counterpart
Methasterone was never a commercially available prescription drug. Its non-17α-alkylated counterpart, drostanolone, was commercialized by Syntex Corporation under the brand name Masteron.[4]
"Designer steroid"
Methasterone resurfaced in 2005 as a “designer steroid”.[5] It was brought to market by Anabolic Xtreme as the primary ingredient of a dietary supplement named Superdrol. Its introduction into commerce may have represented an attempted circumvention of the 1990 Anabolic Steroid Control Act (along with its 2004 revision), since the law is, in part, drug-specific;[6] methasterone, as is the case with many designer steroids, was not declared a Schedule III class anabolic steroid in that act because it was not commercially available at the time the act, and its subsequent revision, were signed into law.[7] Methasterone was therefore being sold as an over-the-counter dietary supplement.
Controversy and FDA involvement
It was in late 2005 that the status of methasterone (Superdrol), in addition to that of four other designer steroids, as a genuine anabolic steroid was brought to public awareness by an article published in the Washington Post.[8] Don Catlin of the UCLA Olympic Laboratory, who conducted the studies, noted methasterone’s similarity to drostanolone. A warning by the FDA was issued soon after to the general public as well as to the distributor, Designer Supplements LLC, for the marketing of this compound.[9] Methasterone was subsequently added to the World Anti-Doping Agency’s list of prohibited substances in sport.[10]. Despite all of this, methasterone (Superdrol) has resurfaced within the supplement industry on several accusations since its banning by WADA [11]
Anabolic efficacy
The anabolic efficacy of this compound can be attested to anecdotally on the many forums online where anabolic drug use practices are openly discussed, as can its hepatoxicity (toxicity to the liver). Many cases of liver damage due to the use of methasterone have been cited in the medical literature.[12] [13] [14] [15] Although the original manufacturer ceased production in the face of possible governmental action, methasterone remains commercially available through over-the-counter dietary supplement products sometimes referred to as “Superdrol clones” and through domestic or foreign chemical production companies.
References
- ^ H. J. Ringold and G. Rosenkranz. “Steroids. LXXXIII. Synthesis of 2-Methyl and 2,2-Dimethyl Hormone Analogs.” Journal of Organic Chemistry. 21. (1956): 1333.
- ^ Ringold, H. J., E. Batres, O. Halpern, and E. Necoechea. Journal of the American Chemical Society. “Steroids. CV.1 2-Methyl and 2-Hydroxymethylene-androstane Derivatives.” 81 (2). (1959): 427-432.
- ^ Julius A. Vida. Androgens and Anabolic Agents: Chemistry and Pharmacology. New York: Academic Press, 1969. 23 & 168.
- ^ “Superdrol, masteron en oxy komen uit hetzelfde nest” [Superdrol, Masteron, and Oxy come from the same nest]. Ergogenics.org. Accessed February 2009. http://www.ergogenics.org/157.html
- ^ Van Enoo, Peter and Frans T. Delbeke. “Metabolism and excretion of anabolic steroids in doping control—New steroids and new insights.” Journal of Steroid Biochemistry & Molecular Biology. 101 (2006): 173.
- ^ Office of Divesion Control, Drug Enforcement Administration, Department of Justice. “Implementation of the Anabolic Steroid Control Act of 2004.” Accessed February 2009. http://www.deadiversion.usdoj.gov/fed_regs/rules/2005/fr1216.htm
- ^ Shipley, Amy, Bonnie Berkowitz and Christina Rivero. “Designer Steroids Hide and Seek.” The Washington Post. October 18, 2005. Accessed February 2009. http://www.washingtonpost.com/wp-dyn/content/graphic/2005/10/18/GR2005101800648.html
- ^ Amy Shipley. “Steroids Detected in Dietary Tablets.” The Washington Post. November 30, 2005. Accessed February 2009. http://www.washingtonpost.com/wp-dyn/content/article/2005/11/29/AR2005112901636.html
- ^ U.S. Food and Drug Administration. “FDA Warns Manufacturers About Illegal Steroid Products Sold as Dietary Supplements” [Press Release]. March 9, 2006. Accessed February 2009. http://www.fda.gov/bbs/topics/NEWS/2006/NEW01332.html
- ^ World Anti-Doping Agency. “The World Anti-Doping Code: The 2009 Prohibited List: International Standard.” Accessed February 2009. http://www.wada-ama.org/rtecontent/document/2009_Prohibited_List_ENG_Final_20_Sept_08.pdf
- ^ http://sportsillustrated.cnn.com/vault/article/magazine/MAG1155395/5/index.htm
- ^ Jasiurkowski, Beata, Jaya Raj, David Wisinger, Richard Carlson, Lixian Zou, and Abdul Nadir. “Cholestatic Jaundice and IgA Nephropathy Induced by OTC Muscle Building Agent Superdrol.” American Journal of Gastroenterology. 101.11 (2006): 2659-2662.
- ^ Nasr, John and Jawad Ahmad. “Severe Cholestasis and Renal Failure Associated with the Use of the Designer Steroid Superdrol (Methasteron): A Case Report and Literature Review.” Digestive Diseases and Sciences. 10.1007 (2008).
- ^ L. Shah, Neeral, Isabel Zacharias, Urmila Khettry, Nezam Afdhal, and Fredric D. Gordon.” Methasteron-Associated Cholestatic Liver Injury: Clinicopathologic Findings in 5 Cases.” Clinical Gastroenterology and Hepatology. 6.2 (2008): 255-258.
- ^ Singh V, Rudraraju M, Carey EJ, Byrne TJ, Vargas HE, Williams JE, Balan V, Douglas DD, Rakela J. “Severe Hepatotoxicity Caused by a Methasteron-containing, Performance-enhancing Supplement.” Journal of Clinical Gastroenterology. Volume 43, Number 3, March (2009).
Categories:- Anabolic steroids
Wikimedia Foundation. 2010.
