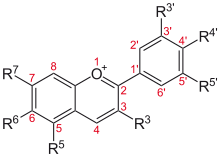
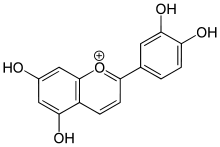
- 3-Deoxyanthocyanidin
-
The 3-Deoxyanthocyanidins and their glycosides (3-deoxyanthocyanins or 3-DA) are molecules with an anthocyanidins backbone lacking an hydroxyl group on carbon 3.
3-Deoxyanthocyanidins are yellow anthocyanidins that can be found primarily in ferns and mosses (Timberlake and Bridle, 1975, 1980)[1], in Sorghum bicolor[1][2] and in purple corn (Nakatani et al., 1979)[1] (maíz morado).
3-Deoxyanthocyanidins are reported to be stable to color loss due to change in pH[3]. Synthetic 3-deoxyanthocyanidins with a carboxylate group at carbon 4 show unusually stable colorant properties at pH 7[1].
In Sorghum, the SbF3'H2 gene, encoding a flavonoid 3'-hydroxylase, seems to be expressed in pathogen-specific 3-deoxyanthocyanidin phytoalexins synthesis[4], for example in Sorghum-Colletotrichum interactions[5].
This category include :
- Apigeninidin
- Columnidin
- Diosmetinidin
- Luteolinidin
- Tricetinidin
References
- ^ a b c d Effect of substitution on the stability of 3-deoxyanthocyanidins in aqueous solutions, James G. Sweeny, Guillermo A. Iacobucci, 1983
- ^ Inclusions of flavonoid 3-deoxyanthocyanidins in Sorghum bicolor self-organize into spherical structures, NIELSEN Kirsten A.; GOTFREDSEN Charlotte H.; BUCH-PEDERSEN Morten J.; AMMITZBØLL Henriette; MATTSSON Ole; DUUS Jens Ø.; NICHOLSON Ralph L, 1986
- ^ Behavior of 3-deoxyanthocyanidins in the presence of phenolic copigments, Joseph M. Awika, 2008
- ^ Differential Expression of Two Flavonoid 3'-Hydroxylase cDNAs Involved in Biosynthesis of Anthocyanin Pigments and 3-Deoxyanthocyanidin Phytoalexins in Sorghum, Chun-Hat Shih, Ivan K. Chu, Wing Kin Yip and Clive Lo, 2006
- ^ Biosynthesis and regulation of 3-deoxyanthocyanidin phytoalexins induced during Sorghum-Colletotrichum interaction: Heterologous expression in maize. Chopra, Surinder Gaffoor, Iffa Ibraheem, Farag
3-hydroxyanthocyanidins 5-Desoxy-peonidin | Aurantinidin | Cyanidin | 6-Hydroxycyanidin | Delphinidin | Fisetinidin | Guibourtinidin | Pelargonidin | Robinetinidin3-deoxyanthocyanidins Apigeninidin | Columnidin | Diosmetinidin | Gesneridin | Luteolinidin | TricetinidinO-methylated anthocyanidins Anthocyanins Antirrhinin | Chrysanthemin | Malvin | Myrtillin | Oenin | Primulin | Pulchellidin 3-glucoside | Pulchellidin 3-rhamnoside | TulipaninAcetylated glycosides Cyanidin-3-(di-p-coumarylglucoside)-5-glucoside (found in Dark opal basil) Violdelphin (found in Aconitum chinense)Misc. Metalloanthocyanins (Commelinin | Cyanosalvianin | Protocyanin | Protodelphin) | Pyranoanthocyanins | CopigmentationCategories:- Anthocyanidins
Wikimedia Foundation. 2010.