- Acetylation
-
Acetylation (or in IUPAC nomenclature ethanoylation) describes a reaction that introduces an acetyl functional group into a chemical compound. (Deacetylation is the removal of the acetyl group.)
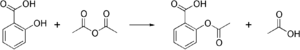
Specifically, acetylation refers to that process of introducing an acetyl group (resulting in an acetoxy group) into a compound, to be specific, the substitution of an acetyl group for an active hydrogen atom. A reaction involving the replacement of the hydrogen atom of a hydroxyl group with an acetyl group (CH3 CO) yields a specific ester, the acetate. Acetic anhydride is commonly used as an acetylating agent reacting with free hydroxyl groups. For example, it is used in the synthesis of aspirin and heroin.
Contents
Acetylation of proteins
In biology, i.e., in living cells, acetylation occurs as a co-translational and post-translational modification of proteins, for example, histones, p53, and tubulins.
N-alpha-terminal Acetylation
Acetylation of the N-terminal alpha-amine of proteins is a widespread modification in eukaryotes. Forty to fifty percent of yeast proteins, and 80-90% of human proteins are modified in this manner, and the pattern of modification is found to be conserved throughout evolution. The modification is performed by N-alpha-acetyltransferases (NATs), a sub-family of the GNAT superfamily of acetyltransferases, which also include histone acetyl transferases. The GNATs transfer the acetylgroup from acetyl-coenzyme A to the amine group. The NATs have been most extensively studied in yeast. Here, three NAT complexes, NatA/B/C, have been found to perform most N-alpha-terminal acetylations. They have sequence specificity for their substrates, and it is believed that they are associated with the ribosome, where they acetylate the nascent polypeptide chain. In humans, the human NatA and NatB complexes have been identified and characterized. Subunits of the human NatA complex have been coupled to cancer-related processes such as hypoxia-response and the beta-catenin pathway. It has been found to be over-expressed in papillary thyroid carcinoma and neuroblastoma. The human NatB complex have been coupled to cell cycle. The hNat3 subunit of the hNatB complex has been found overexpressed in some forms of cancer.
Despite being a conserved and widespread modification, little is known about the biological role of N-alpha-terminal acetylation. Proteins such as actin and tropomyosin have been found to be dependent of NatB acetylation to form proper actin filaments. Yet this is only one example pointing to the potential importance of this modification.
For unknown reasons 20% of Asians have an isozyme that results in slower N-acetylation of drugs, while 50% of Whites and African-Americans do.[citation needed]
Lysine Acetylation and Deacetylation
In histone acetylation and deacetylation, the histones are acetylated and deacetylated on lysine residues in the N-terminal tail as part of gene regulation. Typically, these reactions are catalyzed by enzymes with "histone acetyltransferase" (HAT) or "histone deacetylase" (HDAC) activity, although HATs and HDACs can modify the acetylation status of non-histone proteins as well.[1]
The regulation of transcription factors, effector proteins, molecular chaperones, and cytoskeletal proteins by acetylation/deacetylation is emerging as a significant post-translational regulatory mechanism [2] analogous to phosphorylation by the action of kinases or dephosphorylated by the action of phosphatases. Not only can the acetylation state of a protein modify its activity, there has been recent suggestion that this post-translational modification might crosstalk with phosphorylation, methylation, ubiquitination, sumoylation, and others for dynamic control of cellular signaling.[3]
The tubulin acetylation and deacetylation system is well worked out in Chlamydomonas. A tubulin acetyltransferase located in the axoneme acetylates a specific lysine residue in the α-tubulin subunit in assembled microtubule. Once disassembled, this acetylation can be removed by another specific deacetylase that is cytosolic. Thus the axonemal microtubules (long half-life) carry this signature acetylation absent from cytosolic microtubules (short half-life).
See also
References
- ^ Sadoul K, Boyault C, Pabion M, Khochbin S (February 2008). "Regulation of protein turnover by acetyltransferases and deacetylases". Biochimie 90 (2): 306–12. doi:10.1016/j.biochi.2007.06.009. PMID 17681659.
- ^ Glozak MA, Sengupta N, Zhang X, Seto E (2005). "Acetylation and deacetylation of non-histone proteins". Gene 363: 15–23. doi:10.1016/j.gene.2005.09.010. PMID 16289629.
- ^ Yang XJ, Seto E (2008). "Lysine acetylation: codified crosstalk with other posttranslational modifications". Mol Cell 31 (4): 449–61. doi:10.1016/j.molcel.2008.07.002. PMC 2551738. PMID 18722172. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=2551738.
Protein primary structure and posttranslational modifications General N terminus C terminus Single specific AAs Phosphorylation · Sulfation · Porphyrin ring linkage · Adenylylation · Flavin linkage · Topaquinone (TPQ) formationAspartateGlutamateTransglutaminationMethylation · Acetylation · Acylation · Adenylylation · Hydroxylation · Ubiquitination · Sumoylation · ADP-ribosylation · Deamination · Oxidative deamination to aldehyde · O-glycosylation · Imine formation · Glycation · CarbamylationDiphthamide formation · AdenylylationCrosslinks between two AAs Sulfilimine bondLysine-TyrosylquinoneLysine tyrosylquinone (LTQ) formationTryptophan-TryptophylquinoneThree consecutive AAs
(Chromophore formation)4-(p-hydroxybenzylidene)-5-imidazolinone formationCrosslinks between four AAs Secondary structure→Categories:
Wikimedia Foundation. 2010.