- Paper chromatography
-
Paper chromatography 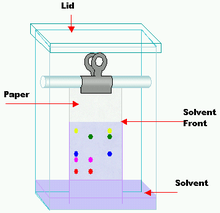
Chromatography jarClassification Chromatography Other techniques Related Thin layer chromatography Paper chromatography is an analytical chemistry technique for separating and identifying mixtures that are or can be colored, especially pigments. This can also be used in secondary or primary colors in ink experiments. This method has been largely replaced by thin layer chromatography, however it is still a powerful teaching tool. Two-way paper chromatography, also called two-dimensional chromatography, involves using two solvents and rotating the paper 90° in between. This is useful for separating complex mixtures of similar compounds, for example, amino acids.
Rƒ value
The retention factor (Rƒ) may be defined as the ratio of the distance traveled by the substance to the distance traveled by the solvent. Rƒ values are usually expressed as a fraction of two decimal places but it was suggested by Smith that a percentage figure should be used instead. If Rƒ value of a solution is zero, the solute remains in the stationary phase and thus it is immobile. If Rƒ value = 1 then the solute has no affinity for the stationary phase and travels with the solvent front. To calculate the Rƒ value, take the distance traveled by the substance divided by the distance traveled by the solvent (as mentioned earlier in terms of ratios). For example, if a compound travels 2.1 cm and the solvent front travels 2.8 cm, (2.1/2.8) the Rƒ value = 0.75
Pigments and polarity
Paper chromatography is one method for testing the purity of compounds and identifying substances. Paper chromatography is a useful technique because it is relatively quick and requires small quantities of material. Separations in paper chromatography involve the same principles as those in thin layer chromatography. In paper chromatography, like thin layer chromatography, substances are distributed between a stationary phase and a mobile phase. The stationary phase is usually a piece of high quality filter paper. The mobile phase is a developing solution that travels up the stationary phase, carrying the samples with it. Components of the sample will separate readily according to how strongly they adsorb on the stationary phase versus how readily they dissolve in the mobile phase.
When a colored chemical sample is placed on a filter paper, the colors separate from the sample by placing one end of the paper in a solvent. The solvent diffuses up the paper, dissolving the various molecules in the sample according to the polarities of the molecules and the solvent. If the sample contains more than one color, that means it must have more than one kind of molecule. Because of the different chemical structures of each kind of molecule, the chances are very high that each molecule will have at least a slightly different polarity, giving each molecule a different solubility in the solvent. The unequal solubilities cause the various color molecules to leave solution at different places as the solvent continues to move up the paper. The more soluble a molecule is, the higher it will migrate up the paper. If a chemical is very nonpolar it will not dissolve at all in a very polar solvent. This is the same for a very polar chemical and a very nonpolar solvent.
Chromatography Techniques Affinity chromatography · Column chromatography · Displacement Chromatography · Electrochromatography · Gas chromatography · High-performance liquid chromatography · Ion chromatography · Micellar electrokinetic chromatography · Normal-phase chromatography · Paper chromatography · Reversed-phase chromatography · Size-exclusion chromatography · Thin-layer chromatography · Two-dimensional chromatographyHyphenated methods Gas chromatography–mass spectrometry · Liquid chromatography–mass spectrometry · Pyrolysis–gas chromatography–mass spectrometryTheory Prominent publications Biomedical Chromatography · Journal of Chromatographic Science · Journal of Chromatography A · Journal of Chromatography B · Journal of Liquid Chromatography & Related Technologies · Journal of Separation ScienceCategories:
Wikimedia Foundation. 2010.
