- Oxazolidine
-
Oxazolidine 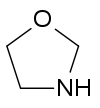 oxazolidineOther names1,3-oxazolidine
oxazolidineOther names1,3-oxazolidineIdentifiers CAS number 504-76-7 Jmol-3D images Image 1 - C1COCN1
Properties Molecular formula C3H7NO Molar mass 73.0938 g/mol  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Oxazolidine is a five-membered ring compound consisting of three carbons, a nitrogen, a hydrogen, and an oxygen. The oxygen and NH are the 1 and 3 positions, respectively. In oxazolidine derivatives, there is always a carbon between the oxygen and the nitrogen (or it would be an isoxazolidine).[1][2] All of the carbons in oxazolidines are reduced (compare to oxazole and oxazoline). Some of their derivatives, the oxazolidinediones, are used as anticonvulsants.
Contents
Bisoxazolidines
Bisoxazolidines have two oxazolidine rings, and they are used as reactive diluents in polyurethane paints. The rings hydrolyze in the presence of moisture into amine and hydroxyl groups, which can then bind with diisocyanates to form the coating.[3]
Isoxazolidines
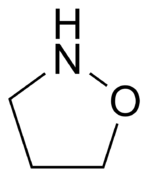
In an isoxazolidine nitrogen and oxygen occupy positions 1 and 2 in the ring:
It is the saturated relative of Isoxazole.
See also
- Imidazole, an analog with the oxygen replaced by a nitrogen.
- Thiazole, an analog with the oxygen replaced by a sulfur.
- Benzoxazole, where the oxazole is fused to another aromatic ring.
- Pyrrole, an analog without the oxygen atom.
- Furan, an analog without the nitrogen atom.
- Oxazoline, which has only one double bond reduced.
- Oxazolidinedione, which has two in-cycle keto groups (a carbamate and a lactam).
- Oxazolidinone, which has an in-cycle carbamate.
References
- ^ "SID 3881507 -- PubChem Substance Summary". The PubChem Project. United States National Center for Biotechnology Information. http://pubchem.ncbi.nlm.nih.gov/summary/summary.cgi?sid=3881507. Retrieved 13 December 2005.
- ^ Dr Neil G Carter OXAZOLIDINE DILUENTS: REACTING FOR THE ENVIRONMENT Industrial Copolymers Limited
- ^ Emission control chembytes e-zine 2001.
Categories:- Oxazolidines
Wikimedia Foundation. 2010.