- Magaldrate
-
Magaldrate 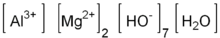
Systematic (IUPAC) name Magnesium aluminate monohydrate Clinical data AHFS/Drugs.com Micromedex Detailed Consumer Information MedlinePlus a682683 Pregnancy cat. C Legal status OTC Routes Oral Pharmacokinetic data Bioavailability Negligible Metabolism Nil Excretion Fecal and renal Identifiers CAS number 1317-26-6 ATC code A02AD02 A02AF01 PubChem CID 6336542 ChemSpider 4891688 
UNII 6V88E24N5T 
Chemical data Formula AlMg2(OH)7·H2O Mol. mass 212.66 g/mol  (what is this?) (verify)
(what is this?) (verify)Magaldrate (INN), is a common antacid drug that is used for the treatment of duodenal and gastric ulcers, esophagitis from gastroesophageal reflux.
Contents
Available forms
Magaldrate is available in the form of oral suspension or tablets.
Interactions and adverse reactions
Magaldrate may negatively influence drugs like tetracyclines, benzodiazepines, and indomethacin. High doses or prolonged usage may lead to an increment of defecation and a reduction in feces consistence. In some cases it can alter the functionality of the gastrointestinal tract, occasionally provoking constipation or diarrhea.
Brand names
The drug is sold with the following brand names: Gadral (Italy); Magaltop (Italy); Magralibi (Italy); Mylanta (Australia); Riopan (Argentina, Austria, Brazil, Germany, Italy, Mexico, Belgium, Switzerland, Greece).
References
Drugs for acid related disorders: Antacids (A02A) → Magnesium
(increases motility)Aluminium
(decreases motility)Calcium Sodium Combinations and complexes
of aluminium, calcium and magnesium
This drug article relating to the gastrointestinal system is a stub. You can help Wikipedia by expanding it.
