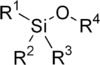- Silyl ether
-
Silyl ethers are a group of chemical compounds which contain a silicon atom covalently bonded to an alkoxy group. The general structure is R1R2R3Si−O−R4 where R4 is an alkyl group or an aryl group. Silyl ethers are usually used as protecting groups for alcohols in organic synthesis. Since R1R2R3 can be combinations of differing groups which can be varied in order to provide a number of silyl ethers, this group of chemical compounds provides a wide spectrum of selectivity for protecting group chemistry. Common silyl ethers are: trimethylsilyl (TMS), tert-butyldiphenylsilyl (TBDPS), tert-butyldimethylsilyl (TBS/TBDMS) and triisopropylsilyl (TIPS), [2-(trimethylsilyl)ethoxy]methyl (SEM). They are particularly useful because they can be installed and removed very selectively under mild conditions.
Contents
Formation
Although many methods are available for forming silyl ethers, there are two common strategies: reaction of the alcohol with a silyl chloride with an amine base at room temperature and reaction of the alcohol with a silyl triflate with a hindered amine base at low temperature. Silyl triflates are more reactive than their corresponding chlorides, so they can be used to install silyl groups onto hindered positions. One extremely reliable and rapid procedure is the Corey protocol in which the alcohol is reacted with a silyl chloride and imidazole at high concentration in DMF.[1] If DMF is replaced by dichloromethane, the reaction is somewhat slower, but the purification of the compound is simplified. A common hindered base for use with silyl triflates is 2,6-lutidine.[2] Primary alcohols can be protected in less than one hour while some hindered alcohols may require days of reaction time.
When using a silyl chloride, no special precautions are usually required, except for the exclusion of large amounts of water. An excess of silyl chloride can be employed but is not necessary. If excess reagent is used, the product will require flash chromatography to remove excess silanol and siloxane. Silyl triflates are water sensitive and must be run under inert atmosphere conditions. Purification involves the addition of an aqueous acid such as saturated ammonium chloride solution. This quenches remaining silyl reagent and protonates amine bases, removing them from the reaction mixture. Following extraction, the product can be purified by flash chromatography.
Silyl triflate is more reactive and also converts ketones to silyl enol ethers.
Removal of silyl ether protecting groups
Reaction with acids or fluorides such as tetra-n-butylammonium fluoride remove the silyl group when protection is no longer needed.[3] Larger substituents increase resistance to hydrolysis, but also make introduction of the silyl group more difficult.[4]
In acidic media, the relative stability is (see above references):
- TMS (1) < TES (64) < TBS (20 000) < TIPS (700 000) < TBDPS (5 000 000)
In basic media, the relative stability is (see above references):
- TMS (1) < TES (10-100) < TBS~TBDPS (20 000) < TIPS (100 000)
Monoprotection of symmetrical diols
It is possible to monosilylate a symmetrical diol, although this is known to be problematic occasionally. For example, the following monosilylation was reported:[5]
However, it turns out that this reaction is hard to repeat. If the reaction were controlled solely by thermodynamics then, statistically, if the dianion is of similar reactivity to the monoanion, then a corresponding statistical mixture of 1:2:1 disilylated:monosilylated:unsilylated diol will result. However, the reaction in THF is made selective by two factors kinetic deprotonation of the first anion AND the insolubility of the monoanion. At the initial addition of TBSCl, there is only a minor amount of monoanion in solution with the rest being in suspension. This small portion reacts and shifts the equilibrium of the monoanion to draw more into solution, thereby allowing for high yields of the mono-TBS compound to be obtained. Superior results in some cases may obtained with butyllithium:[6]
A third method uses a mixture of DMF and DIPEA.[7]
Alternatively, an excess (4 eq) of the diol can be used, forcing the reaction toward monoprotection.
Selective deprotection
Selective deprotection of silyl groups is possible in many instances. For example:[8]
Silyl ethers are mainly differentiated on the basis of sterics or electronics. In general, acidic deprotections deprotect less hindered silyl groups faster, with the steric bulk on silicon being more significant than the steric bulk on oxygen. Fluoride-based deprotections deprotect electron-poor silyl groups faster than electron-rich silyl groups. There is some evidence that some silyl deprotections proceed via hypervalent silicon species.
The selective deprotection of silyl ethers has been extensively reviewed.[9][10] Although selective deprotections have been achieved under many different conditions, some procedures, outlined below, are more reliable. A selective deprotection will likely be successful if there is a substantial difference in sterics (e.g., primary TBS vs. secondary TBS or primary TES vs primary TBS) or electronics (e.g. primary TBDPS vs. primary TBS). Unfortunately, some optimization is inevitably required and it is often necessary to run deprotections partway and recycle material.
Some Common Acidic Conditions:
- 100 mol% 10-CSA (camphorsulfonic acid) in MeOH, room temperature; a "blast" of acid, deprotects primary TBS groups within ten minutes.
- 10 mol% 10-CSA, 1:1 MeOH:DCM, −20 or 0 °C; deprotects a primary TBS group within two hours at 0; if CSA is replaced by PPTS, the rate is approximately ten times slower; with pTsOH, approximately ten times faster; solvent mixture is crucial.
- 4:1:1 v/v/v AcOH:THF:water, room temp.; this is very slow, but can be very selective.
Some Common Basic Conditions:
- HF-pyridine, 10:1 THF:pyridine, 0 °C; an excellent deprotection; removes primary TBS groups within eight hours; reactions using HF must be run in plastic containers.
- TBAF, THF or 1:1 TBAF/AcOH, THF; TBDPS and TBS groups can be deprotected in the presence of one another under different conditions.[11]
References
- ^ Corey, E. J.; Venkateswarlu, A. "Protection of hydroxyl groups as tert-butyldimethylsilyl derivatives." J. Am. Chem. Soc. 1972, 94, 6190–6191. doi:10.1021/ja00772a043
- ^ Corey, E. J.; Cho, H.; Rücker C.; Hua, D. H. "Studies with trialkylsilyltriflates: new syntheses and applications." Tetrahedron Lett. 1981, 22, 3455–3458. doi:10.1016/S0040-4039(01)81930-4
- ^ Greene, T. W.; Wuts, P. G. M. Protective Groups In Organic Synthesis, 3rd ed.; John Wiley & Sons: New York, 1991.
- ^ Greene, T. W.; Wuts, P. G. M. Protective Groups In Organic Synthesis, 3rd ed.; John Wiley & Sons: New York, 1991.
- ^ McDougal, P. G.; Rico, J. G.; Oh, Y.-I.; Condon, B. D. "A convenient procedure for the monosilylation of symmetric 1,n-diols." J. Org. Chem. 1986, 51, 3388–3390. doi:10.1021/jo00367a033
- ^ Roush, W. R.; Gillis, H. R.; Essenfeld, A. P. "Hydrofluoric acid catalyzed intramolecular Diels-Alder reactions " J. Org. Chem. 1983, 49, 4674-4682. doi:10.1021/jo00198a018
- ^ Hu, L.; Liu, B.; Yu, C. Tetrahedron Lett. 2000, 41, 4281. doi:10.1016/S0040-4039(00)00626-2
- ^ Holton, R. A. et al. "First total synthesis of taxol. 2. Completion of the C and D rings." J. Am. Chem. Soc. 1994, 116, 1599–1600. doi:10.1021/ja00083a067
- ^ Nelson, T. D.; Crouch, R. D. "Selective deprotection of silyl ethers." Synthesis 1996, 1031–1069. doi:10.1055/s-1996-4350
- ^ Crouch, R. D. "Selective monodeprotection of bis-silyl ethers." Tetrahedron 2004, 60, 5833–5871. doi:10.1016/j.tet.2004.04.042
- ^ Higashibayashi, S.; Shinko, K.; Ishizu, T.; Hashimoto, K.; Shirahama, H.; Nakata, M. "Selective deprotection of t-butyldiphenylsilyl ethers in the presence of t-butyldimethylsilyl ethers by tetrabutylammonium fluoride, acetic acid, and water." Synlett 2000, 1306–1308. doi:10.1055/s-2000-7158
External links
Categories:- Functional groups
- Protecting groups
Wikimedia Foundation. 2010.