- Chloro(triphenylphosphine)gold(I)
-
Chloro(triphenylphosphine)gold(I) 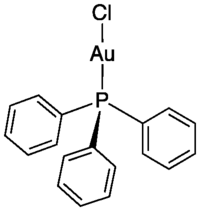
Identifiers CAS number !14243-64-2 Properties Molecular formula C18H15AuClP Molar mass 494.71 g mol−1 Appearance Colorless solid Melting point 236-237 °C
 (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Chloro(triphenylphosphine)gold(I) is the coordination complex with the formula (Ph3P)AuCl (Ph = phenyl). This colorless solid is the complex of triphenylphosphine and gold(I) chloride.
Preparation and structure
The complex is prepared by reducing chloroauric acid with triphenylphosphine in 95% ethanol:[1]
- HAuCl4 + H2O + 2 PPh3 → (Ph3P)AuCl + Ph3PO + 3 HCl
The complex adopts a linear coordination geometry, which is typical of most gold(I) compounds.[2][3]
Reactivity
It is a common reagent in gold chemistry. Many related gold complexes (often incorrectly called salts) can be prepared including the trifluoromethanesulfonate and the nitrate.[4]
Triphenylphosphine gold triflate (Ph3PAuOTf) catalyzes certain rearrangement reactions in organic synthesis.
References
- ^ Pierre Braunstein, Hans Lehner, Dominique Matt "A Platinum-Gold Cluster: Chloro-1κCl-Bis(Triethylphosphine-1κP)Bis(Triphenyl- Phosphine)-2κP, 3κP-Triangulo- Digold-Platinum(1 +) Trifluoromethanesulfonate" Inorganic Syntheses, 1990, Volume 27, Pages 218–221. doi: 10.1002/9780470132586.ch42
- ^ . doi:10.1107/S0567740876004330.
- ^ . doi:10.1021/jp807258d.
- ^ A. M. Mueting, B. D. Alexander, P. D. Boyle, A. L. Casalnuovo, L. N. Ito, B. J. Johnson, L. H. Pignolet "Mixed-Metal-Gold Phosphine Cluster Compounds" Inorganic Syntheses, 1992, Volume 29, Pages 279–298, 2007. doi:10.1002/9780470132609.ch63
Categories:- Coordination compounds
- Gold compounds
- Chemistry stubs
Wikimedia Foundation. 2010.
