- Chloroacetone
-
Chloroacetone 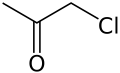 ChloropropanoneOther namesAcetonyl chloride, chloropropanone, 1-chloro-2-propanone, monochloroacetone, 1-chloro-2-ketopropane, 1-chloro-2-oxypropane
ChloropropanoneOther namesAcetonyl chloride, chloropropanone, 1-chloro-2-propanone, monochloroacetone, 1-chloro-2-ketopropane, 1-chloro-2-oxypropane
UN 1695Identifiers CAS number 78-95-5 
PubChem 6571 ChemSpider 6323 
UNII 60ZTR74268 
ChEBI CHEBI:47220 
RTECS number UC0700000 Jmol-3D images Image 1 - ClCC(=O)C
Properties Molecular formula C3H5ClO Molar mass 92.52 g mol−1 Appearance Colorless liquid, oxidizes to amber Density 1.15 g/cm3 at 20°C Melting point -44.5 °C, 229 K, -48 °F
Boiling point 119 °C, 392 K, 246 °F
Solubility in water 10 g/100 mL at 20 °C Solubility alcohol, ether, chloroform[1] Vapor pressure 1.5 kPa Hazards Flash point 35 °C Autoignition
temperature610 °C Explosive limits 3.4% - ?[2] LD50 100 mg/kg (rats, oral)[3]  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Chloroacetone is a chemical compound with the formula CH3COCH2Cl. At STP it is a colourless liquid with a pungent odour.[4] On exposure to light, it turns to a dark yellow-amber colour.[5] It was used as a tear gas in World War I.[6]
Contents
Applications
Chloroacetone is used to make dye couplers for colour photography, and is an intermediate in chemical manufacturing[3]. It is also used in the Feist-Benary synthesis of furans[7].

See also
- Use of poison gas in World War I
- Bromoacetone
References
- ^ Lide, David R. (1998). Handbook of Chemistry and Physics (87 ed.). Boca Raton, FL: CRC Press. pp. 3–100. ISBN 0849305942.
- ^ "ICSC:NENG0760 International Chemical Safety Cards (WHO/IPCS/ILO) CDC/NIOSH". Center for Disease Control. 2006-10-11. http://www.cdc.gov/niosh/ipcsneng/neng0760.html. Retrieved 2009-04-17.
- ^ a b Hathaway, Gloria J.; Proctor, Nick H. (2004). Proctor and Hughes' Chemical Hazards of the Workplace (5 ed.). Wiley-Interscience. pp. 143–144. ISBN 9780471268833. http://books.google.com/?id=y3-Ef3y53PkC&pg=PA143&dq=Chloroacetone. Retrieved 2009-04-16.
- ^ "Occupational Safety and Health Guideline for Chloroacetone". U.S. Department of Labor - Occupational Safety & Health Administration. http://www.osha.gov/SLTC/healthguidelines/chloroacetone/recognition.html. Retrieved 2008-06-06.
- ^ "CHLOROACETONE". International Programme on Chemical Safety. http://www.inchem.org/documents/icsc/icsc/eics0760.htm. Retrieved 2008-06-06.
- ^ Haber, Ludwig Fritz (1986). The Poisonous Cloud: Chemical Warfare in the First World War. Oxford University press. ISBN 0198581424.
- ^ Li, Jie-Jack; Corey, E. J. (2004). Name Reactions in Heterocyclic Chemistry. Wiley-Interscience. pp. 160. ISBN 9780471302155. http://books.google.com/?id=1N-MZVSesTcC&pg=PA160&dq=Chloroacetone. Retrieved 2009-04-16.
External links
This article about an organic compound is a stub. You can help Wikipedia by expanding it.
