- Diamine
-
A diamine is a type of polyamine with exactly two amino groups. Diamines are mainly used as monomers to prepare polyamides, polyimides and polyureas. In terms of quantities produced, 1,6-diaminohexane, a precursor to Nylon 6-6, is most important, followed by ethylenediamine.[1] Hydrazine (H2NNH2) is usually not considered a diamine since it is neither an amine nor is it dibasic.
Contents
Aliphatic diamines
Linear aliphatic diamines
- 2 carbons: ethylenediamine (1,2-diaminoethane). Related derivatives include the N-alkylated compounds ethambutol and TMEDA.
- 3 carbons: 1,3-diaminopropane (propane-1,3-diamine)
- 4 carbons: putrescine (butane-1,4-diamine)
- 5 carbons: cadaverine (pentane-1,5-diamine)
- 6 carbons: hexamethylenediamine (hexane-1,6-diamine)
Branched aliphatic diamines
Derivatives of ethylenediamine are numerous:
- 1,2-diaminopropane
- diphenylethylenediamine
- diaminocyclohexane
Xylylenediamines
Xylylenediamines are classified as alkylamines since the amine is not directly attached to an aromatic ring.
- o-xylylenediamine or OXD
- m-xylylenediamine or MXD
- p-xylylenediamine or PXD
Aromatic diamines
Three phenylenediamines are known:[2]
- o-phenylenediamine or OPD
- m-phenylenediamine or MPD
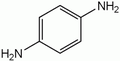
- p-phenylenediamine or PPD. 2,5-Diaminotoluene is related to PPD but contains a methyl group on the ring.
Various N-methylated derivatives of the phenylenediamines are known:
- dimethyl-4-phenylenediamine, a reagent.
- N,N'-di-2-butyl-1,4-phenylenediamine, an antioxidant.
Examples with two aromatic rings include derivatives of biphenyl and naphthalene:
-
PPD (para-phenylenediamine)
References
- ^ Karsten Eller, Erhard Henkes, Roland Rossbacher, Hartmut Höke "Amines, Aliphatic" in Ullmann's Encyclopedia of Industrial Chemistry, 2005 Wiley-VCH Verlag, Weinheim. doi:10.1002/14356007.a02_001
- ^ Robert A. Smiley “Phenylene- and Toluenediamines” in Ullmann's Encyclopedia of Industrial Chemistry 2002, Wiley-VCH, Weinheim. doi:10.1002/14356007.a19_405
External links

This article about an amine is a stub. You can help Wikipedia by expanding it.