- Dhurrin
-
Dhurrin 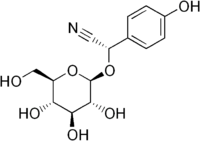 (2S)-2-(4-Hydroxyphenyl)-2-[[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)-2-tetrahydropyranyl]oxy]acetonitrileOther names(S)-4-Hydroxymandelnitrile-β-D-glucopyranoside; (S)-(β-D-Glucopyranosyloxy)(4-hydroxyphenyl)acetonitrile
(2S)-2-(4-Hydroxyphenyl)-2-[[(2R,3R,4S,5S,6R)-3,4,5-trihydroxy-6-(hydroxymethyl)-2-tetrahydropyranyl]oxy]acetonitrileOther names(S)-4-Hydroxymandelnitrile-β-D-glucopyranoside; (S)-(β-D-Glucopyranosyloxy)(4-hydroxyphenyl)acetonitrileIdentifiers CAS number 499-20-7 PubChem 161355 Jmol-3D images Image 1 - C1=CC(=CC=C1[C@@H](C#N)O[C@H]2[C@@H]([C@H]([C@@H]([C@H](O2)CO)O)O)O)O
Properties Molecular formula C14H17NO7 Molar mass 311.29 g/mol  (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Dhurrin is a cyanogenic glycoside occurring in plants. Its biosynthesis has been elucidated.[1][2] Dhurrin is hydrolyzed in the stomach of an insect into a carbohydrate and aglycone. The aglycone is unstable and releases hydrogen cyanide.
References
- ^ Nielsen JS, Moller BL (1999). "Biosynthesis of cyanogenic glucosides in Triglochin maritima and the involvement of cytochrome P450 enzymes". Arch. Biochem. Biophys. 368 (1): 121–130. doi:10.1006/abbi.1999.1258. PMID 10415119.
- ^ Nielsen JS, Moller BL (2000). "Cloning and expression of cytochrome P450 enzymes catalyzing the conversion of tyrosine to p-hydroxyphenylacetaldoxime in the biosynthesis of cyanogenic glucosides in Triglochin maritima". Plant Physiol. 122 (4): 1311. doi:10.1104/pp.122.4.1311. PMC 58967. PMID 10759528. http://www.pubmedcentral.nih.gov/articlerender.fcgi?tool=pmcentrez&artid=58967.
Bond Geometry Glycone Aglycone biochemical families: prot · nucl · carb (glpr, alco, glys) · lipd (fata/i, phld, strd, gllp, eico) · amac/i · ncbs/i · ttpy/i Categories:- Glucosides
- Nitriles
- Phenols
- Biochemistry stubs
Wikimedia Foundation. 2010.
