- Spinosad
-
Spinosyn A 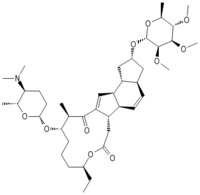
Identifiers CAS number 131929-63-0 PubChem 183094 ChemSpider 16736513 
ChEMBL CHEMBL1615373 ATCvet code QP53 - InChI=1S/C42H67NO10.C41H65NO10/c1-11-26-13-12-14-35(53-37-16-15-34(43(6)7)24(4)49-37)23(3)38(45)33-20-31-29(32(33)21-36(44)51-26)17-22(2)28-18-27(19-30(28)31)52-42-41(48-10)40(47-9)39(46-8)25(5)50-42;1-10-26-12-11-13-34(52-36-17-16-33(42(5)6)23(3)48-36)22(2)37(44)32-20-30-28(31(32)21-35(43)50-26)15-14-25-18-27(19-29(25)30)51-41-40(47-9)39(46-8)38(45-7)24(4)49-41/h17,20,23-32,34-35,37,39-42H,11-16,18-19,21H2,1-10H3;14-15,20,22-31,33-34,36,38-41H,10-13,16-19,21H2,1-9H3/t23-,24-,25+,26+,27-,28+,29-,30-,31-,32+,34+,35+,37+,39+,40-,41-,42+;22-,23-,24+,25-,26+,27-,28-,29-,30-,31+,33+,34+,36+,38+,39-,40-,41+/m11/s1

Key: JFLRKDZMHNBDQS-SGSTVUCESA-N
InChI=1/C42H67NO10.C41H65NO10/c1-11-26-13-12-14-35(53-37-16-15-34(43(6)7)24(4)49-37)23(3)38(45)33-20-31-29(32(33)21-36(44)51-26)17-22(2)28-18-27(19-30(28)31)52-42-41(48-10)40(47-9)39(46-8)25(5)50-42;1-10-26-12-11-13-34(52-36-17-16-33(42(5)6)23(3)48-36)22(2)37(44)32-20-30-28(31(32)21-35(43)50-26)15-14-25-18-27(19-29(25)30)51-41-40(47-9)39(46-8)38(45-7)24(4)49-41/h17,20,23-32,34-35,37,39-42H,11-16,18-19,21H2,1-10H3;14-15,20,22-31,33-34,36,38-41H,10-13,16-19,21H2,1-9H3/t23-,24-,25+,26+,27-,28+,29-,30-,31-,32+,34+,35+,37+,39+,40-,41-,42+;22-,23-,24+,25-,26+,27-,28-,29-,30-,31+,33+,34+,36+,38+,39-,40-,41+/m11/s1
Key: JFLRKDZMHNBDQS-SGSTVUCEBI
 (verify) (what is:
(verify) (what is:  /
/ ?)
?)
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa)Infobox references Spinosyn D 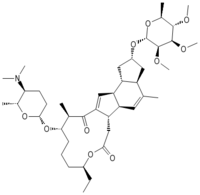
Identifiers CAS number 131929-60-7 PubChem 443059 ATCvet code QP53 Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox references Spinosad Combination of Spinosyn A ? Class Spinosyn D ? Class Clinical data AHFS/Drugs.com International Drug Names MedlinePlus a611012 Licence data US FDA:link Pregnancy cat. B(US) Legal status ℞-only (US) Routes Topical Identifiers ATC code ? UNII XPA88EAP6V Spinosad (spinosyn A and spinosyn D) is a new chemical class of insecticides that are registered by the United States Environmental Protection Agency (EPA) to control a variety of insects. The active ingredient is derived from a naturally occurring soil dwelling bacterium called Saccharopolyspora spinosa, a rare actinomycete reportedly collected from soil in an abandoned rum distillery on a Caribbean Island in 1982 by a scientist on vacation.[1] It has not been found in nature since that time, and was subsequently described as a new species. The bacteria produce compounds (metabolites) while in a fermentation broth. The first fermentation-derived compound was formulated in 1988. Spinosad has since been formulated into insecticides that combine the efficacy of a synthetic insecticide with the benefits of a biological pest control organism.
Spinosad topical suspension, 0.9%, was approved by the U.S. Food and Drug Administration (FDA) on 2011-01-18 for the topical treatment of head lice infestation in patients four (4) years of age and older.[2] It is marketed as the trade name Natroba in the US by ParaPRO Pharmaceuticals.[2] Spinosad is a mixture of spinosyn A and spinosyn D in a ratio of approximately 5 to 1 (spinosyn A to spinosyn D).[3]
Contents
Mode of action
Spinosad kills susceptible species by causing rapid excitation of the insect nervous system. Due to this unique mode of action, Spinosad is valued in resistance management programs. Spinosad must be ingested by the insect, therefore it has little effect on non-target predatory insects. It is highly effective used as an oral anti-flea medication for dogs, and apparently has the potential for killing any blood-sucking insect.
Spinosad is relatively fast acting. The insect dies within one to two days after ingesting the active ingredient. There appears to be 100% mortality.
Use
It is used to control a variety of insect pests, including fruit flies, caterpillars, leafminers, thrips, sawflies, spider mites, fire ants, and leaf beetle larvae. Spinosad is recommended for use in an Integrated Pest Management program for commercial greenhouses since it will not harm most beneficial insects or predatory mites. Spinosad does not significantly affect beneficial organisms including ladybugs, green lacewings, minute pirate bugs, and predatory mites. It has also been tested, with promising results, as a mosquito-control agent in West Africa and Israel.[4]
Products
Spinosad has been formulated into two commercial products registered for use in commercial agriculture, including DowAgros' Conserve SC, and Entrust. Entrust is approved for use on USDA certified organic produce.
There are several home garden products manufactured for use in the United States including Monterey Garden Insect Spray manufactured by Lawn and Garden Products, Inc. and 'Borer, Bagworm, Tent Caterpillar & Leafminer Spray' manufactured by Ferti-Lome. Bonide Products has introduced spinosad in its Captain Jacks Dead Bug Brew insect control product line, and offers another product, "Colorado Potato Beetle Beater", with spinosad as its active ingredient.
The products have practically no odor. Its soil absorption is moderately strong and it degrades rapidly in the environment primarily through photolysis.
Spinosad is also the active ingredient in Comfortis,[5] a chewable flea medication for dogs.[5]
Safety
Its "Caution" signal word indicates a reduced risk to applicators and workers. There are no specific worker protection requirements, even though applicators and handlers should wear a long-sleeved shirt, long pants, shoes and socks.
Spinosad shows low toxicity when ingested by mammals (male rat LD50 = 3738 mg/kg) and no additional adverse effects from chronic exposure. Studies on spinosad show slight toxicity to birds, moderate toxicity to fish, and slight to moderate toxicity to aquatic invertebrates. However, it is highly toxic to bees (honey bee LC50 = 11.5 ppm) and is highly toxic to oysters and other marine mollusks. Applications to areas where bees are actively foraging should be avoided. After the residues have dried, it is much less toxic to bees.
It is important to note that toxicity is based on the active ingredient tested; formulations of spinosad in common use today have a very small amount of spinosad active ingredient. In addition, non-target sensitivity is mitigated by the environmental characteristics of spinosad, including rapid dissipation in the water column, sorption and binding of residues with sediment and lack of bioaccumulation in living tissues.
Beneficial Trichogramma and Braconid wasps are harmed by the chemical, but it has much less effect on such insects as lacewings and beetles like the ladybug and spares predatory bugs such as damsel bugs and the big-eyed bug.[6]
Spinosad does not have any phytotoxicity on ornamentals[citation needed] and is non-phytotoxic for most crops.[citation needed] It has a 4-hour Worker Protection Standard reentry interval (REI).[citation needed]
Do not apply Spinosad more than 10 times in a 12-month period inside a greenhouse to prevent possible insect pesticide resistance.[citation needed]
Sources
- Dow AgroSciences LLC. Conserve SC InfoSheet. Indianapolis IN.
- "A review of Spinosad" Tam Tran. University of California, Irvine.
- Extension Toxicology Network. Pesticide Information Profiles. EPA Pesticide Programs.
- Bio-Itegral Resource Center.
References
- ^ http://www.dowagro.com/PublishedLiterature/dh_005f/0901b8038005f580.pdf
- ^ a b Natroba Drug Approval Package. U.S. Food and Drug Administration (FDA).
- ^ Natroba full prescribing information label. U.S. Food and Drug Administration (FDA).
- ^ McNeil, Jr., Donald (26 September 2011). "Brewing Up Double-Edged Delicacies for Mosquitoes". New York Times. http://www.nytimes.com/2011/09/27/health/27mosquito.html?_r=1&sq=mosquitoes&st=cse&adxnnl=1&scp=2&adxnnlx=1317243706-57bt9L1o6rSenb9VjAESNg. Retrieved 28 September 2011.
- ^ a b Comfortis label. Elanco Animal Health. Eli Lilly.
- ^ USDA fact sheet[dead link]
Categories:- Biological pest control
- Insecticides
- Organic gardening
- Sustainable agriculture
- InChI=1S/C42H67NO10.C41H65NO10/c1-11-26-13-12-14-35(53-37-16-15-34(43(6)7)24(4)49-37)23(3)38(45)33-20-31-29(32(33)21-36(44)51-26)17-22(2)28-18-27(19-30(28)31)52-42-41(48-10)40(47-9)39(46-8)25(5)50-42;1-10-26-12-11-13-34(52-36-17-16-33(42(5)6)23(3)48-36)22(2)37(44)32-20-30-28(31(32)21-35(43)50-26)15-14-25-18-27(19-29(25)30)51-41-40(47-9)39(46-8)38(45-7)24(4)49-41/h17,20,23-32,34-35,37,39-42H,11-16,18-19,21H2,1-10H3;14-15,20,22-31,33-34,36,38-41H,10-13,16-19,21H2,1-9H3/t23-,24-,25+,26+,27-,28+,29-,30-,31-,32+,34+,35+,37+,39+,40-,41-,42+;22-,23-,24+,25-,26+,27-,28-,29-,30-,31+,33+,34+,36+,38+,39-,40-,41+/m11/s1
Wikimedia Foundation. 2010.
