- Nitrogen-13
-
Nitrogen-13 General Name, symbol Nitrogen-13,13N Neutrons 6 Protons 7 Nuclide data Half-life <10 minutes Parent isotopes 13O (Electron capture) Decay mode Decay energy Electron capture Nitrogen-13 is a radioisotope of nitrogen used in positron emission tomography (PET). It has a half life of a little under ten minutes, so it must be made at the PET site. A cyclotron may be used for this purpose.
Nitrogen-13 is used to tag ammonia molecules for PET.
Contents
Production
- H1 + O16 → N13 + He4
The proton must be accelerated to a kinetic energy of about 5.55 Mev or a little more.
The reaction is endothermic (i.e. the mass of the products is greater than the reactants, so energy needs to be supplied which is converted to mass).This is one reason why the proton needs to carry extra energy to produce the nuclear reaction.
The energy difference is actually 5.22 MeV, but if the proton only supplied this energy the reactants would be formed with no kinetic energy. As momentum must be conserved, the true energy that needs to be supplied by the proton is given by:
- K = (1 + m / M) | E |
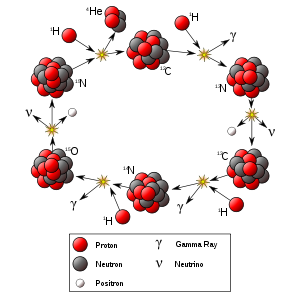
Role in stellar fusion
Nitrogen-13 plays a significant role in the CNO cycle, which is the dominant source of energy in stars heavier than the sun.[1]
-
12
6C+ 1
1H→ 13
7N+ γ + 1.95 MeV 13
7N→ 13
6C+ e+
+ ν
e+ 2.22 MeV 13
6C+ 1
1H→ 14
7N+ γ + 7.54 MeV 14
7N+ 1
1H→ 15
8O+ γ + 7.35 MeV 15
8O→ 15
7N+ e+
+ ν
e+ 2.75 MeV 15
7N+ 1
1H→ 12
6C+ 4
2He+ 4.96 MeV
External links
- PET site of the University of Melbourne
References
Lighter:
Nitrogen-12Nitrogen-13 is an
isotope of NitrogenHeavier:
Nitrogen-14Decay product of:
Oxygen-13 (electron capture)Decay chain
of Nitrogen-13Decays to:
Carbon-13 (EC)Categories:
Wikimedia Foundation. 2010.