- Converting (metallurgy)
-
Converting is a term used to describe a number of metallurgical smelting processes. The most commercially important use of the term is in the treatment of molten metal sulfides to produce crude metal and slag, as in the case of copper and nickel converting. Another, now uncommon, use of the term referred to batch treatment of pig iron to produce steel by the Bessemer process. The vessel used was called the Bessemer converter.
Converting in copper metallurgy
A mixture of copper and iron sulfides referred to as matte is treated in converters to oxidize iron in the first stage, and oxidize copper in the second stage. In the first stage oxygen enriched air is blown through the tuyeres to partially convert metal sulfides to oxides:
- FeS + O2 → FeO + SO2
- CuS + O2 → CuO + SO2
Since iron has greater affinity to oxygen, the produced copper oxide reacts with the remaining iron sulfide:
- CuO + FeS → CuS + FeO
The bulk of the copper oxide is turned back into the form of sulfide. In order to separate the obtained iron oxide, flux (mainly silica) is added into the converter. Silica reacts with iron oxide to produce a light slag phase, which is poured off through the hood when the converter is tilted around the rotation axis:
- 2 FeO + SiO2 → Fe2SiO4 (sometimes denoted as 2FeO•SiO2, fayalite)
After the first portion of slag is poured off the converter, a new portion of matte is added, and the converting operation is repeated many times until the converter is filled with the purified copper sulfide. The converter slag is usually recycled to the smelting stage due to the high content of copper in this by-product. Converter gas contains more than 10% of sulfur dioxide, which is usually captured for the production of sulfuric acid.
The second stage of converting is aimed at oxidizing the copper sulfide phase (purified in the first stage), and produces blister copper. The following reaction takes place in the converter:
- CuS + O2 → Cu + SO2
Copper content in the obtained blister copper is typically more than 95%. Blister copper is the final product of converting.
Equipment
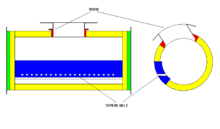
The converting process occurs in a converter. Two kinds of converters are widely used: horizontal and vertical.
Horizontal converters of the Pierce-Smith type prevail in the metallurgy of non ferrous metals. Such a converter is a horizontal barrel lined with refractory material inside. A hood for the purpose of the loading/unloading operations is located on the upper side of the converter. Two belts of tuyeres come along the axis on either sides of the converter.
Molten sulfide material, referred to as matte, is poured through the hood into the converter during the operation of loading. Air is distributed to tuyeres from the two tuyere collectors which are located on opposite sides of the converter. Collector pipes vary in diameter with distance from the connection to air supplying trunk; this is to provide equal pressure of air in each tuyere.
See also
Categories:- Metallurgical processes
Wikimedia Foundation. 2010.