- Rieske protein
Pfam_box
Symbol = Rieske
Name =
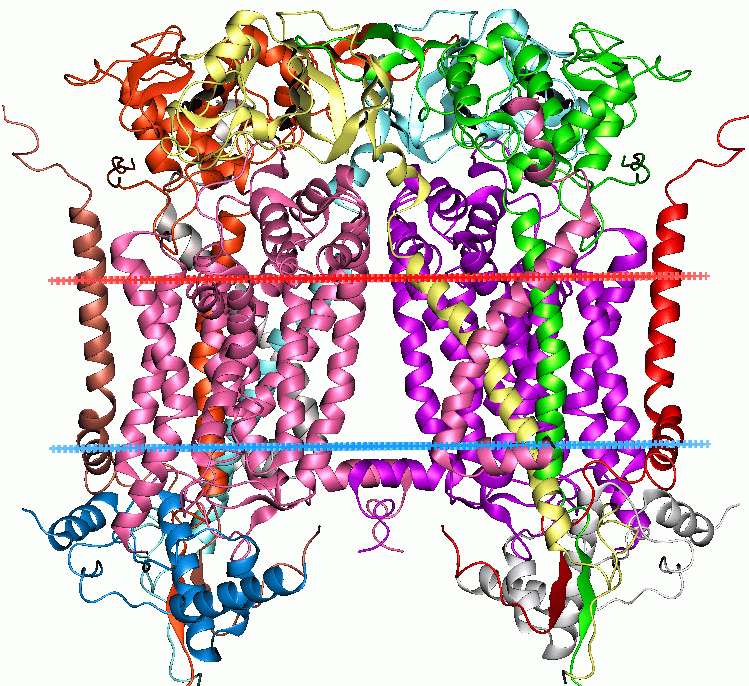
width =260
caption =Mitochondrialcytochrome bc1 complex . (PDB|1bcc)
Pfam= PF00355
InterPro= IPR005806
SMART=
PROSITE= PDOC00177
SCOP = 1rie
TCDB = 3.E.2
OPM family=
OPM protein= 1q90
PDB=PDB3|1ww9A:28-131 PDB3|1z01E:43-146 PDB3|1z03C:43-146PDB3|1z02A:43-146 PDB3|1fqtA:2-96 PDB3|1vckA:4-98PDB3|1sjgA:2-96 PDB3|1vm9A:2-96 PDB3|1wqlA:58-165PDB3|1uliA:55-148 PDB3|1uljC:55-148 PDB3|2bmqA:36-137PDB3|2bmoA:36-137 PDB3|2bmrA:36-137 PDB3|1o7nA:38-139PDB3|1o7wA:38-139 PDB3|1eg9A:38-139 PDB3|1o7hA:38-139PDB3|1ndoC:38-139 PDB3|1o7pA:38-139 PDB3|1uuvA:38-139PDB3|1o7mA:38-139 PDB3|1uuwA:38-139 PDB3|1o7gA:38-139PDB3|2b1xA:45-158 PDB3|2b24A:45-158 PDB3|1rfs :137-206PDB3|1q90C:113-182 PDB3|1vf5Q:64-158 PDB3|2d2cQ:64-158PDB3|1g8kH:47-110 PDB3|1g8jD:47-110 PDB3|1nykA:89-185PDB3|1jm1A:96-204 PDB3|1kyoP:116-208 PDB3|1p84E:116-208PDB3|1kb9E:116-208 PDB3|1nu1E:174-266 PDB3|1rie :174-266PDB3|1ntzE:174-266 PDB3|1sqqE:174-266 PDB3|1l0nE:174-266PDB3|1be3E:174-266 PDB3|1sqvE:174-266 PDB3|1sqpE:174-266PDB3|1l0lE:174-266 PDB3|1bccE:174-266 PDB3|1ntmE:174-266PDB3|1ntkE:174-266 PDB3|1sqbE:174-266 PDB3|1sqxE:174-266Rieske protein is a
iron-sulfur protein (ISP) component of cytochrome "bc"1 complex which was first discovered and isolated by John S. Rieske and co-workers in 1964.Biological function
Ubiquinol-cytochrome-c reductase (also known as bc1 complex or complex III) is an enzyme complex of bacterial and mitochondrial oxidative phosphorylation systems It catalyses the oxidoreduction of the mobile redox componentsubiquinol andcytochrome c , generating anelectrochemical potential , which is linked to ATP synthesiscite journal |author=Harnisch U, Weiss H, Sebald W |title=The primary structure of the iron-sulfur subunit of ubiquinol-cytochrome c reductase from Neurospora, determined by cDNA and gene sequencing |journal=Eur. J. Biochem. |volume=149 |issue=1 |pages=95–99 |year=1985 |pmid=2986972 |doi=10.1111/j.1432-1033.1985.tb08898.x] cite journal |author=Sebald W, Gabellini N |title=Nucleotide sequence and transcription of the fbc operon from Rhodopseudomonas sphaeroides. Evaluation of the deduced amino acid sequences of the FeS protein, cytochrome b and cytochrome c1 |journal=Eur. J. Biochem. |volume=154 |issue=3 |pages=569–579 |year=1986 |pmid=3004982 |doi=10.1111/j.1432-1033.1986.tb09437.x] . The complex consists of three subunits in most bacteria, and nine in mitochondria: both bacterial and mitochondrial complexes contain cytochrome b and cytochrome c1 subunits, and an iron-sulphur 'Rieske' subunit, which contains a high potential 2Fe-2S clustercite journal |author=Ludwig B, Kurowski B |title=The genes of the Paracoccus denitrificans bc1 complex. Nucleotide sequence and homologies between bacterial and mitochondrial subunits |journal=J. Biol. Chem. |volume=262 |issue=28 |pages=13805–13811 |year=1987 |pmid=2820981] .The mitochondrial form also includes six other subunits that do not possess redox centres. Plastoquinone-plastocyanin reductase (b6f complex), present in cyanobacteria and the chloroplasts of plants, catalyses the oxidoreduction of plastoquinol and cytochrome f. This complex, which is functionally similar to ubiquinol-cytochrome c reductase, comprises cytochrome b6, cytochrome f and Rieske subunitscite journal |author=Madueno F, Napier JA, Cejudo FJ, Gray JC |title=Import and processing of the precursor of the Rieske FeS protein of tobacco chloroplasts |journal=Plant Mol. Biol. |volume=20 |issue=2 |pages=289–299 |year=1992 |pmid=1391772 |doi=10.1007/BF00014496] .The Rieske subunit acts by binding either a
ubiquinol orplastoquinol anion, transferring an electron to the 2Fe-2S cluster, then releasing the electron to thecytochrome c orcytochrome f haem iron. The rieske domain has a [2Fe-2S] centre. Two conserved cysteines coordinate one Fe ion while the other Fe ion is coordinated by two conserved histidines. The 2Fe-2S cluster is bound in the highly conserved C-terminal region of the Rieske subunit.Rieske protein family
The homologues of the Rieske proteins include ISP components of cytochrome "b"6"f" complex,
aromatic-ring-hydroxylating dioxygenases (phthalate dioxygenase, benzene, napthalene and toluene 1,2-dioxygenases) and arsenite oxidase (EC [http://www.expasy.org/cgi-bin/get-enzyme-entry?1.20.98.1 1.20.98.1] ). Comparison of amino acid sequences has revealed the following consensus sequence:: Cys-Xaa-His-(Xaa)15–17-Cys-Xaa-Xaa-His3D structure
The crystal structures of a number of Rieske proteins are known. The overall fold, comprising two subdomains, is dominated by antiparallel β-structure and contains the only α-helix. The smaller "cluster-binding" subdomains in mitochondrial and chloroplast proteins are virtually identical, whereas the large subdomains are substantially different in spite of a common folding topology. The [Fe2S2] cluster-binding subdomains have the topology of an incomplete antiparallel β-barrel. One iron atom of the Rieske [Fe2S2] cluster is coordinated by two
cysteine residues and the other is coordinated by twohistidine residues through the Nδ atoms. The ligands coordinating the cluster originate from two loops; each loop contributes one Cys and one His.ubfamilies
*
Rieske iron-sulphur protein, C-terminal InterPro|IPR005805
*Arsenite oxidase, small subunit InterPro|IPR014067Human proteins containing this domain
AIFM3 ;RFESD ;UQCRFS1 ;References
Further reading
*Structure of a water soluble fragment of the 'Rieske' iron- sulfur protein of the bovine heart mitochondrial cytochrome bc1 complex determined by MAD phasing at 1.5 A resolution. Iwata S, Saynovits M, Link TA, Michel H Structure 1996;4:567-579. PMID|8736555
*Functional analysis in yeast of cDNA coding for the mitochondrial Rieske iron-sulfur protein of higher plants. Huang JT, Struck F, Matzinger DF, Levings CS; Proc Natl Acad Sci U S A 1991;88:10716-10720. PMID|1961737
*The mitochondrial targeting presequence of the Rieske iron-sulfur protein is processed in a single step after insertion into the cytochrome bc1 complex in mammals and retained as a subunit in the complex. Brandt U, Yu L, Yu CA, Trumpower BL; J Biol Chem 1993;268:8387-8390. PMID|8386158
*
*
*
*
*
*External links
* - X-ray structure of Rieske protein (water-soluble fragment) of the bovine mitochondrial cytochrome "bc"1 complex
* - X-ray structure of Rieske protein (water-soluble fragment) of the spinach chloroplast cytochrome "b"6" f"complex
* - X-ray structure of Rieske-type ferredoxin associated with biphenyl dioxygenase from "Burkholderia cepacia "
* - X-ray structure of Rieske subunit of arsenite oxidase from "Alcaligenes faecalis"
* - InterPro entry for Rieske [2Fe-2S] region
Wikimedia Foundation. 2010.
