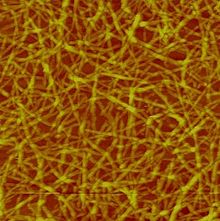
- Nanocellulose
-
Nanocellulose or microfibrillated cellulose (MFC), is a material composed of nanosized cellulose fibrils with a high aspect ratio (length to width ratio). Typical dimensions are 5–20 nanometers width and length up to 2000 nanometers. It is pseudo-plastic. Moreover, nanocellulose exhibits the property of certain gels or fluids that are thick (viscous) under normal conditions, but flow (become thin, less viscous) over time when shaken, agitated, or otherwise stressed. This property is known as thixotropy. When the shearing forces are removed the gel regains much of its original state. The fibrils are isolated from any cellulose containing source including wood-based fibers (pulp fibers) through high-pressure, high temperature and high velocity impact homogenization (see manufacture below). Nanocellulose can also be obtained from native fibers by an acid hydrolysis, giving rise to highly crystalline and rigid nanoparticles (generally referred to as nanowhiskers) which are shorter (100s to 1000 nanometers) than the nanofibrils obtained through the homogenization route.
Contents
History and terminology
The terminology microfibrillated/nanocellulose or (MFC) was first used by Turbak, Snyder and Sandberg in the late 1970s at the ITT Rayonier labs in Whippany, New Jersey, USA to describe a product prepared as a gel type material by passing wood pulp through a Gaulin type milk homogenizer at high temperatures and high pressures followed by ejection impact against a hard surface. The terminology (MFC) first appeared publicly in the early 1980s when a number of patents and publications issued to ITT Rayonier on this totally new nanocellulose composition of matter.[1] In later work Herrick at Rayonier also published work on making a dry powder form of the gel.[2] Since Rayonier is one of the world's premier producer of purified pulps their business interests have always been 1) to create new uses and new markets for pulps and 2) never to compete with new or potentially new customers. Thus, as the patents issued,[3] Rayonier gave free license to whomever wanted to pursue this new use for cellulose. Rayonier,as a company, never pursued scale-up. Rather, Turbak et al. pursued 1) finding new uses for the MFC/nanocellulose. These included using MFC as a thickener and binder in foods, cosmetics, paper formation, textiles, nonwovens, etc. and 2) evaluate swelling and other techniques for lowering the energy requirements for MFC/Nanocellulose production.[4] ITT closed the Rayonier Whippany Labs in 1983–84 and further work on making a dry powder form of MFC was done by Herric at the Rayonier labs in Shelton, Washington, USA[2]
The field was later taken up in Japan in the mid 1990s by the group of Taniguchi and co-workers and later by Yano and co-workers.[5] and a host of major companies (see numerous U.S. patents issued to P&G, J&J, 3M, McNeil, etc. using U.S. patent search under inventor name Turbak search base). Today, there are still extensive research and development efforts around the world in this field.
Manufacture
Nanocellulose/MFC can be prepared from any cellulose source material, but woodpulp is normally used. The nanocellulose fibrils are isolated from the wood-based fibers using high-pressure homogenizers. The homogenizers are used to delaminate the cell walls of the fibers and liberating the nanosized fibrils. This production route is normally connected to high energy consumptions associated with the fiber delamination. Values over 30 MWh/tonne are not uncommon. Pre-treatments are sometimes used to address this problem. Examples of such pre-treatments are enzymatic/mechanical pre-treatment[6] and introduction of charged groups e.g. through carboxymethylation[7] or TEMPO-mediated oxidation. It has been shown that energy consumption can be heavily decreased using these pre-treatments and Lindström and Ankerfors has reported values below 1 MWh/tonne.[8] Cellulose nanowhiskers, a more crystalline form of nanocellulose, is formed by the acid hydrolysis of native cellulose fibers using a concentrated inorganic salt, commonly sulfuric or hydrochloric acid. The amorphous sections of native cellulose are hydrolysed and after careful timing, the crystalline sections can be retrieved from the acid solution by centrifugation and washing. Cellulose nanowhiskers are rodlike highly crystalline particles (relative crystallinity index above 75%) with a rectangular cross section. Its dimensions depend on the native cellulose source material, and hydrolysis time and temperature.
Structure and properties
Nanocellulose dimensions and crystallinity
The ultrastructure of cellulose derived from various sources has been extensively studied. Techniques such as transmission electron microscopy (TEM), scanning electron microscopy (SEM), atomic force microscopy (AFM), wide angle X-ray scattering (WAXS), small incidence angle X-ray diffraction and solid state 13C cross-polarization magic angle spinning (CP/MAS) nuclear magnetic resonance (NMR) spectroscopy have been used to characterize nanocellulose morphology. These methods have typically been applied for the investigation of dried nanocellulose morphology.[9]
Although a combination of microscopic techniques with image analysis can provide information on nanocellulose fibril widths, it is more difficult to determine nanocellulose fibril lengths because of entanglements and difficulties in identifying both ends of individual nanofibrils. It is often reported that nanocellulose suspensions are not homogeneous and that they consist of cellulose nanofibers and nanofiber bundles.
Most methods have typically been applied to investigation of dried nanocellulose dimensions, although a study was conducted where the size and size-distribution of enzymatically pre-treated nanocellulose fibrils in a suspension was studied using cryo-TEM. The fibrils were found to be rather mono-dispersed mostly with a diameter of ca. 5 nm although occasionally thicker fibril bundles were present.[6] It should be noted that, some newly published results indicated that by combining ultrasonication with an "oxidation pretreatment", cellulose microfibrils with a lateral dimension that belows 1 nm is observed by AFM. The lower end of the thickness dimension is around 0.4 nm, which is believed to be the thickness of a cellulose monolayer sheet.[10]
The aggregate widths can be determined by CP/MAS NMR developed by Innventia AB, Sweden, which also has been demonstrated to work for nanocellulose (enzymatic pre-treatment). An average width of 17 nm has been measured with the NMR-method, which corresponds well with SEM and TEM. Using TEM, values of 15 nm have been reported for nanocellulose from carboxymethylated pulp. However, also thinner fibrils can also be detected. Wågberg et al. reported fibril widths of 5–15 nm for a nanocellulose with a charge density of about 0.5 meq./g.[7] The group of Isogai reported fibril widths of 3–5 nm for TEMPO-oxidized cellulose having a charge density of 1.5 meq./g.[11]
The influence of cellulose pulp chemistry on the nanocellulose microstructure has been investigated using AFM to compare the microstructure of two types of nanocellulose prepared at Innventia AB (enzymatically pre-treated nanocellulose and carboxymethylated nanocellulose). Due to the chemistry involved in producing carboxymethylated nanocellulose, it differs significantly from the enzymatically pre-treated one. The number of charged groups on the fibril surfaces are very different. The carboxymethylation pre-treatment makes the fibrils highly charged and, hence, easier to liberate, which results in smaller and more uniform fibril widths (5–15 nm) compared to the enzymatically pre-treated nanocellulose, where the fibril widths were 10–30 nm.[12] The degree of crystallinity and the cellulose crystal structure of nanocellulose were also studied at the same time. The results clearly showed the nanocellulose exhibited cellulose crystal I organization and that the degree of crystallinity was unchanged by the preparation of the nanocellulose. Typical values for the degree of crystallinity were around 63%.[12]
Viscosity
The unique rheology of nanocellulose dispersions was recognized by the early investigators.[13] The high viscosity at low nanocellulose concentrations makes nanocellulose very interesting as a non-calorie stabilizer and gellant in food applications, the major field explored by the early investigators.
The dynamic rheological properties have been investigated in great detail [6] and it has been found that the storage and loss modulus were independent of the angular frequency at all nanocellulose concentrations between 0.125% to 5.9%. The storage modulus values are particularly high (104 Pa at 3% concentration)[6] compared to results for cellulose nanowhiskers (102 Pa at 3% concentration).[13] There is also a particular strong concentration dependence as the storage modulus increases 5 orders of magnitude if the concentration is increased from 0.125% to 5.9%.
Nanocellulose gels are also highly shear thinning (the viscosity is lost upon introduction of the shear forces). The shear-thinning behaviour is, of course, particularly useful in a range of different coating applications.[6]
Mechanical properties
It has long been known that crystalline cellulose has interesting mechanical properties for use in material applications. The stiffness of crystalline cellulose has been shown to be in the order of 140–220 GPa, which is in the same size order as for instance Kevlar and is better than, for example, glass fibers, both fibers are used commercially to reinforce plastics. Films made from nanocellulose have been shown to have high strength (over 200 MPa), high stiffness (around 20 GPa) and high strain (12 %).
Barrier properties
In semi-crystalline polymers, the crystalline regions are considered to be gas impermeable. Due to relatively high crystallinity,[12] in combination with the ability of the nanofibers to form a dense network held together by strong inter-fibrillar bonds (high cohesive energy density), it has been suggested that nanocellulose might act as a barrier material.[11][14][15] Although the number of reported oxygen permeability values are limited, reports attribute high oxygen barrier properties to nanocellulose films. One study reported an oxygen permeability of 0.0006 (cm³ µm)/(m² day kPa) for a ca. 5 µm thin nanocellulose film at 23 °C and 0 % RH.[14] In a related study, a more than 700-fold decrease in oxygen permeability of a polylactide (PLA) film when an nanocellulose layer was added to the PLA surface was reported.[11]
The influence of nanocellulose film density and porosity on film permeability remains relatively unexplored. Some authors have reported significant porosity in nanocellulose films,[16][17][18] which seems to be in contradiction with high oxygen barrier properties, whereas Aulin et al.[14] measured a nanocellulose film density close to density of crystalline cellulose (cellulose Iß crystal structure, 1.63 g/cm³)[19] indicating a very dense film with a porosity close to zero.
Changing the surface functionality of the cellulose nanoparticle can also affect the permeability of nanocellulose films. Films constituted of negatively charged cellulose nanowhiskers could effectively reduce permeation of negatively charged ions, while leaving neutral ions virtually unaffected. Positively charged ions were found to accumulate in the membrane.[20]
Foams
Nanocellulose can also be used to make aerogels/foams, either by itself or in composite formulations. Nanocellulose-based foams are being studied for packaging applications in order to replace polystyrene-based foams. Svagan et al. showed that nanocellulose has the ability to reinforce starch foams by using a freeze-drying technique.[21] The advantage of using nanocellulose instead of wood-based pulp fibers is that the nanofibrills can reinforce the thin cells in the starch foam. Moreover, it is possible to prepare pure nanocellulose aerogels applying various freeze-drying and super critical CO2 drying techniques. Aerogels and foams can be used as porous templates, potentially useful in various nanoapplications.[22][23] Tough ultra-high porosity foams prepared from cellulose I nanofibrill suspensions were studied by Sehaquiet al. A wide range of mechanical properties including compression was obtained by controlling density and nanofibrill interaction in the foams.[24] Cellulose nanowhiskers could also be made to gel in water under low power sonication giving rise to aerogels with the highest reported surface area (>600m2/g) and lowest shrinkage during drying (6.5%) for cellulose aerogels.[23] In another study by Aulin et al.,[25] the formation of structured porous aerogels of nanocellulose by freeze-drying was demonstrated. The density and surface texture of the aerogels was be tuned by selecting the concentration of the nanocellulose dispersions before freeze-drying. Chemical vapour deposition of a fluorinated silane was used to uniformly coat the aerogel to tune their wetting properties towards non-polar liquids/oils. The authors demonstrated that it is possible to switch the wettability behaviour of the cellulose surfaces between super-wetting and super-repellent, using different scales of roughness and porosity created by the freeze-drying technique and change of concentration of the nanocellulose dispersion. Structured porous cellulose foams can however also be obtained by utilizing the freeze-drying technique on cellulose generated by Gluconobacter strains of bacteria, which bio-synthesize open porous networks of cellulose fibers with relatively large amounts of nanofibrills dispersed inside. Olsson et al.[26] demonstrated that these networks can be further impregnated with metalhydroxide/oxide precursors, which can readily be transformed into grafted magnetic nanoparticles along the cellulose nanofibers. The magnetic cellulose foam may allow for a number of novel applications of nanocellulose and the first remotely actuated magnetic super sponges absorbing 1 gram of water within a 60 mg cellulose aerogel foam were reported. Notably, these highly porous foams (>98% air) can be compressed into strong magnetic nanopapers, which may find use as functional membranes in various applications.
Surface modification
The surface modification of nanocellulose is currently receiving a large amount of attention.[27] Nanocellulose displays a large amount of hydroxyl groups at the surface which can be reacted. However, hydrogen bonding strongly affects the reactivity of the surface hydroxyl groups. In addition, impurities at the surface of nanocellulose such as glucosidic and lignin fragments need to be removed before surface modification to obtain acceptable reproducibility between different batches.[28]
Applications
The properties of nanocellulose (e.g. mechanical properties, film-forming properties, viscosity etc.) makes it an interesting material for many applications.
Paper and paperboard
The potential of nanocellulose applications in the area of paper and paperboard manufacture is obvious. Nanocelluloses are expected to enhance the fiber-fiber bond strength and, hence, have a strong reinforcement effect on paper materials. Nanocellulose may be useful as a barrier in grease-proof type of papers and as a wet-end additive to enhance retention, dry and wet strength in commodity type of paper and board products.
Composite
As described above the properties of the nanocellulose makes an interesting material for reinforcing plastics. Nanocellulose has been reported to improve the mechanical properties of e.g. thermosetting resins, starch-based matrixes, soy protein, rubber latex, poly(lactide). The composite applications may be for use as coatings and films, paints, foams, packaging.
Food
Nanocellulose can be used as a low calorie replacement for today’s carbohydrate additives used as thickeners, flavour carriers and suspension stabilizers in a wide variety of food products and is useful for producing fillings, crushes, chips, wafers, soups, gravies, puddings etc. The food applications were early recognised as a highly interesting application field for nanocellulose due to the rheological behaviour of the nanocellulose gel.
Hygiene and absorbent products
Different applications in this field include:
- Super water absorbent (e.g. for incontinence pads material)
- Nanocellulose used together with super absorbent polymers
- Use of nanocellulose in tissue, non-woven products or absorbent structures
- Use as antimicrobial films
Emulsion and dispersion
Apart from the numerous applications in the area of food additives, the general area of emulsion and dispersion applications in other fields has also got some attention. Oil in water applications were early recognized. The area of non-settling suspensions for pumping sand, coal as well as paints and drilling muds was also explored by the early investigators.
Oil recovery
Hydrocarbon fracturing of oil-bearing formations is a potentially interesting and large-scale application. Nanocellulose has been suggested for use in oil recovery applications as a fracturing fluid. Drilling muds based on nanocellulose has also been suggested.
Medical, cosmetic and pharmaceutical
The use of nanocellulose in cosmetics and pharmaceuticals was also early recognized. A wide range of high-end applications have been suggested:
- Freeze-dried nanocellulose aerogels used in sanitary napkins, tampons, diapers or as wound dressing
- The use of nanocellulose as a composite coating agent in cosmetics e.g. for hair, eyelashes, eyebrows or nails
- A dry solid nanocellulose composition in the form of tablets for treating intestinal orders
- Nanocellulose films for screening of biological compounds and nucleic acids encoding a biological compound
- Filter medium partly based on nanocellulose for leukocyte free blood transfusion
- A buccodental formulation, comprising nanocellulose and a polyhydroxylated organic compound
- Powdered nanocellulose has also been suggested as an excipient in pharmaceutical compositions
- nanocellulose in compositions of a photoreactive noxious substance purging agent
Other applications
- Nanocellulose used to activate the dissolution of cellulose in different solvents
- Regenerated cellulose products, such as fibers films, cellulose derivatives
- Tobacco filter additive
- Organometallic modified nanocellulose in battery separators
- Nanocellulose reinforcement of conductive materials
- The use of nanocellulose in loud-speaker membranes
- High-flux membranes
See also
- Cellulose
- Cellulose fiber
- Composite material
References
- ^ Turbak, A.F.; F.W. Snyder, and K.R. Sandberg (1983). "Microfibrillated cellulose, a new cellulose product: Properties, uses and commercial potential". In A. Sarko (ed.). Proceedings of the Ninth Cellulose Conference. Applied Polymer Symposia, 37. New York City: Wiley. pp. 815–827. ISBN 0-471-88132-5.
- ^ a b Herrick, F.W.; R.L. Casebier, J.K. Hamilton, and K.R. Sandberg (1983). "Microfibrillated cellulose: morphology and accessibility". In A. Sarko (ed.). Proceedings of the Ninth Cellulose Conference. Applied Polymer Symposia, 37. New York City: Wiley. pp. 797–813. ISBN 0-471-88132-5.
- ^ Turbak, A.F., F.W. Snyder, and K.R. Sandberg U.S. Patent 4,341,807; U.S. Patent 4,374,702; U.S. Patent 4,378,381; U.S. Patent 4,452,721; U.S. Patent 4,452,722; U.S. Patent 4,464,287; U.S. Patent 4,483,743; U.S. Patent 4,487,634; U.S. Patent 4,500,546
- ^ Turbak, A.F., F.W. Snyder, and K.R. Sandberg “Microfibrillated Cellulose—A New Composition of Commercial Significance,” 1984 Nonwovens Symposium, Myrtle Beach, SC, Apr. 16–19, 1984, pages 115–124./publisher = TAPPI Press, Atlanta, GA
- ^ Berglund, Lars (2005). "Cellulose-based nanocomposites". In A.K. Mohanty, M. Misra, and L. Drzal (Eds). Natural fibers, biopolymers and biocomposites. Boca Raton, Florida: CRC Press. pp. 807–832. ISBN 978-0-8493-1741-5.
- ^ a b c d e Pääkkö, M.; M. Ankerfors, H. Kosonen, A. Nykänen, S. Ahola, M. Österberg, J. Ruokolainen, J. Laine, P.T. Larsson, O. Ikkala, and T. Lindström (2007). "Enzymatic hydrolysis combined with mechanical shearing and high-pressure homogenization for nanoscale cellulose fibrils and strong gels". Biomacromolecules 8 (6): 1934–1941. doi:10.1021/bm061215p. PMID 17474776.
- ^ a b Wågberg, Lars; Gero Decher, Magnus Norgren, Tom Lindström, Mikael Ankerfors, and Karl Axnäs (2008). "The build-up of polyelectrolyte multilayers of microfibrillated cellulose and cationic polyelectrolytes". Langmuir 24 (3): 784–795. doi:10.1021/la702481v. PMID 18186655.
- ^ Lindström, Tom; Mikael Ankerfors (2009). "NanoCellulose Developments in Scandinavia". 7th International Paper and Coating Chemistry Symposium (Preprint CD ed.). Hamilton, Ontario: McMaster University Engineering. ISBN 978-0-981287-90-4.
- ^ Siró, István; David Plackett (2010). "Microfibrillated cellulose and new nanocomposite materials: a review". Cellulose 17 (3): 459–494. doi:10.1007/s10570-010-9405-y.
- ^ Li, Qingqing; Scott Renneckar (06). "Supramolecular Structure Characterization of Molecularly Thin Cellulose I Nanoparticles". Biomacromolecules 12 (3): 650–659. doi:10.1021/bm101315y. http://pubs.acs.org/doi/abs/10.1021/bm101315y. Retrieved 19 March 2011.
- ^ a b c Fukuzumi, Hayaka; Tsuguyuki Saito, Tadahisa Iwata, Yoshiaki Kumamoto and Akira Isogai (2009). "Transparent and high gas barrier films of cellulose nanofibers prepared by TEMPO-mediated oxidation". Biomacromolecules 10 (1): 162–165. doi:10.1021/bm801065u. PMID 19055320.
- ^ a b c Aulin, Christian; Susanna Ahola, Peter Josefsson, Takashi Nishino, Yasuo Hirose, Monika Österberg and Lars Wågberg (2009). "Nanoscale Cellulose Films with Different Crystallinities and Mesostructures-Their Surface Properties and Interaction with Water". Langmuir 25 (13): 7675–7685. doi:10.1021/la900323n. PMID 19348478.
- ^ a b Tatsumi, Daisuke; Satoshi Ishioka and Takayoshi Matsumoto (2002). "Effect of Fiber Concentration and Axial Ratio on the Rheological Properties of Cellulose Fiber Suspensions". Journal of the Society of Rheology (Japan) 30 (1): 27–32. doi:10.1678/rheology.30.27. http://www.jstage.jst.go.jp/article/rheology/30/1/27/_pdf.
- ^ a b c Aulin, Christian; Mikael Gällstedt and Tom Lindström (2010). "Oxygen and oil barrier properties of microfibrillated cellulose films and coatings". Cellulose 17 (3): 559–574. doi:10.1007/s10570-009-9393-y.
- ^ Syverud, Kristin; Per Stenius (2009). "Strength and barrier properties of MFC films". Cellulose 16 (1): 75–85. doi:10.1007/s10570-008-9244-2.
- ^ Henriksson, Marielle; Lars Berglund (2007). "Structure and properties of cellulose nanocomposite films containing melamine formaldehyde". Journal of Applied Polymer Science 106 (4): 2817–2824. doi:10.1002/app.26946. http://intra.kth.se/polopoly_fs/1.23850!SAPOC_MH.pdf.
- ^ Svagan, Anna J.; My A. S. Azizi Samir, and Lars A. Berglund (2007). "Biomimetic polysaccharide nanocomposites of high cellulose content and high toughness". Biomacromolecules 8 (8): 2556–2563. doi:10.1021/bm0703160. PMID 17655354.
- ^ Henriksson, Marielle; Lars A. Berglund, Per Isaksson, Tom Lindström and Takashi Nishino (2008). "Cellulose nanopaper structures of high toughness". Biomacromolecules 9 (6): 1579–1585. doi:10.1021/bm800038n. PMID 18498189.
- ^ Diddens, Imke; Bridget Murphy, Michael Krisch and Martin Müller (2008). "Anisotropic elastic properties of cellulose measured using inelastic x-ray scattering". Macromolecules 41 (24): 9755–9759. Bibcode 2008MaMol..41.9755D. doi:10.1021/ma801796u.
- ^ Thielemans, Wim; Warbey, C.A, Walsh, D.A. (2009). "Permselective nanostructured membranes based on cellulose nanowhiskers". Green Chemistry 11: 531–537. doi:10.1039/b818056c.
- ^ Svagan, Anna J.; My A. S. Azizi Samir, Lars A. Berglund (2008). "Biomimetic foams of high mechanical performance based on nanostructured cell walls reinforced by native nanofibrils". Advanced Materials 20 (7): 1263–1269. doi:10.1002/adma.200701215.
- ^ Pääkkö, Marjo; Jaana Vapaavuori, Riitta Silvennoinen, Harri Kosonen, Mikael Ankerfors, Tom Lindström, Lars A. Berglund and Olli Ikkala (2008). "Long and entangled nantive cellulose I nanofibers allow flexible aerogels and hierarchically templates for functionalities". Soft Matter 4: 2492–2499. doi:10.1039/b810371b.
- ^ a b Heath, Lindy; Thielemans, W. (2010). "Cellulose nanowhisker aerogels". Green Chemistry 12: 1448–1453. doi:10.1039/c0gc00035c.
- ^ Sehaqui, Houssine; Michaela Salajková, Qi Zhou and Lars A. Berglund (2010). "Mechanical performance tailoring of tough ultra-high porosity foams prepared from cellulose I nanofiber suspensions". Soft Matter 6: 1824–1832. doi:10.1039/b927505c.
- ^ Aulin, Christian; Julia Netrval, Lars Wågberg and Tom Lindström (2010). "Aerogels from nanofibrillated cellulose with tunable oleophobicity". Soft Matter 6 (Advance publication): 3298. doi:10.1039/c001939a.
- ^ Olsson, R. T.; Azizi Samir, M. A. S.; Salazar-Alvarez, G.; Belova, L.; Str�m, V.; Berglund, L. A.; Ikkala, O.; Nogu�s, J. et al. (2010). "Making flexible magnetic aerogels and stiff magnetic nanopaper using cellulose nanofibrils as templates". Nature Nanotechnology 5 (8): 584. Bibcode 2010NatNa...5..584O. doi:10.1038/nnano.2010.155.
- ^ Eichhorn, S.J.; A. Dufresne, M. Aranguren, N. E. Marcovich, J. R. Capadona, S. J. Rowan, C. Weder, W. Thielemans, M. Roman, S. Renneckar, W. Gindl, S. Veigel, J. Keckes, H. Yano, K. Abe, M. Nogi, A. N. Nakagaito, A. Mangalam, J. Simonsen, A. S. Benight, A. Bismarck, L. A. Berglund, T. Peijs (2010). "Review: current international research into cellulose nanofibres and nanocomposites". Journal of Materials Science 45: 1–33. doi:10.1007/s10853-009-3874-0.
- ^ Labet, M.; Thielemans, W (2011). "Improving the reproducibility of chemical reactions on the surface of cellulose nanocrystals: ROP of e-caprolactone as a case study". Cellulose 18: 607–617. doi:10.1007/s10570-011-9527-x.
Categories:- Polymers
- Cellulose
- Nanotechnology
Wikimedia Foundation. 2010.