- Etoricoxib
-
Etoricoxib 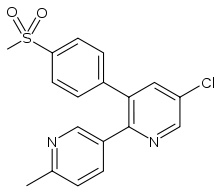
Systematic (IUPAC) name 5-chloro-6'-methyl-3-[4-(methylsulfonyl)phenyl]-
2,3'-bipyridineClinical data AHFS/Drugs.com International Drug Names Pregnancy cat. Not recommended Legal status POM (UK) Routes Oral Pharmacokinetic data Bioavailability 100% Protein binding 92% Metabolism Hepatic, CYP extensively involved (mainly CYP3A4) Half-life 22 hours Excretion Renal (70%) and fecal (20%) Identifiers CAS number 202409-33-4 
ATC code M01AH05 PubChem CID 123619 DrugBank DB01628 ChemSpider 110209 
UNII WRX4NFY03R 
KEGG D03710 
ChEBI CHEBI:6339 
ChEMBL CHEMBL416146 
Chemical data Formula C18H15ClN2O2S Mol. mass 358.842 g/mol SMILES eMolecules & PubChem  (what is this?) (verify)
(what is this?) (verify)Etoricoxib (brand name'NUSHINMASCOT HEALTH SERIES Arcoxia worldwide; also Algix and Tauxib in Italy, Etosaid in India) is a COX-2 selective inhibitor (approx. 106.0 times more selective for COX-2 inhibition over COX-1) from Merck & Co. Currently it is approved in more than 70 countries worldwide but not in the US, where the Food and Drug Administration (FDA) requires additional safety and efficacy data for etoricoxib before it will issue approval. Current therapeutic indications are: treatment of rheumatoid arthritis, psoriatic arthritis, osteoarthritis, ankylosing spondylitis, chronic low back pain, acute pain and gout. Note that approved indications differ by country.
Like any other COX-2 selective inhibitor ("coxib"), etoricoxib selectively inhibits isoform 2 of the enzyme cyclo-oxigenase (COX-2). This reduces the generation of prostaglandins (PGs) from arachidonic acid. Among the different functions exerted by PGs, their role in the inflammation cascade should be highlighted. COX-2 selective inhibitors showed less marked activity on type 1 cycloxigenase compared to traditional non-steroidal anti-inflammatory drugs (NSAID). This reduced activity is the cause of reduced gastrointestinal side effects, as demonstrated in several large clinical trials performed with different coxibs.[1][2]
Some clinical trials and meta-analysis showed that treatment with some coxibs (in particular rofecoxib) led to increased incidence of adverse cardiovascular events compared to placebo. Because of these results, some drugs were withdrawn from the market (rofecoxib, in September 2004 and valdecoxib in April 2005). In addition, the FDA and EMA (USA and European Community health authorities respectively) started a revision process of the entire class of both NSAID and COX-2 inhibitors.
The FDA concluded its revision on April 6, 2005: the final document can be found here.
The EMA concluded its revision on June 27, 2005: the final document can be found here.
On April 27, 2007, the Food and Drug Administration issued Merck a "non-approvable letter" for etoricoxib. The letter said Merck needs to provide more test results showing that the drug's benefits outweigh its risks before it has another chance of getting approved.
References
Anti-inflammatory products (M01A) Pyrazolidine/Butylpyrazolidines Ampyrone • Clofezone • Kebuzone • Metamizole • Mofebutazone • Oxyphenbutazone • Phenazone • Phenylbutazone • Sulfinpyrazone • Feprazone •Acetic acid derivatives
and related substancesAceclofenac • Acemetacin • Alclofenac • Bromfenac • Bumadizone • Bufexamac • Diclofenac • Difenpiramide • Etodolac • Fentiazac • Indometacin • Ketorolac • Lonazolac • Oxametacin • Proglumetacin • Sulindac • Tolmetin • Zomepirac • AmfenacOxicams Propionic acid derivatives Alminoprofen • Benoxaprofen • Dexibuprofen • Dexketoprofen • Fenbufen • Fenoprofen • Flunoxaprofen • Flurbiprofen • Ibuprofen • Ibuproxam • Indoprofen • Ketoprofen • Naproxen • Oxaprozin • Pirprofen • Suprofen • Tiaprofenic acidFenamates Coxibs Other 
This drug article relating to the musculoskeletal system is a stub. You can help Wikipedia by expanding it.
