- Chlorfenapyr
-
Chlorfenapyr 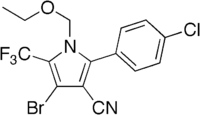 4-Bromo-2-(4-chlorophenyl)-1-ethoxymethyl-5-trifluoromethyl-1H-pyrrole-3-carbonitrile
4-Bromo-2-(4-chlorophenyl)-1-ethoxymethyl-5-trifluoromethyl-1H-pyrrole-3-carbonitrileIdentifiers CAS number 122453-73-0 ChemSpider 82875 UNII NWI20P05EB KEGG C18455 
Jmol-3D images Image 1 - Clc2ccc(c1c(C#N)c(Br)c(n1COCC)C(F)(F)F)cc2
- InChI=1/C15H11BrClF3N2O/c1-2-23-8-22-13(9-3-5-10(17)6-4-9)11(7-21)12(16)14(22)15(18,19)20/h3-6H,2,8H2,1H3
Key: CWFOCCVIPCEQCK-UHFFFAOYAO
Properties Molecular formula C15H11BrClF3N2O Molar mass 407.6 g/mol Density 0.543 g/ml tapped bulk density Melting point 100-101 °C
Except where noted otherwise, data are given for materials in their standard state (at 25 °C, 100 kPa) Infobox references Chlorfenapyr is a pesticide, and specifically a pro-insecticide (meaning it is metabolized into an active insecticide after entering the host), derived from a class of microbially produced compounds known as halogenated pyrroles. The United States Environmental Protection Agency initially denied registration in 2000 for use on cotton primarily because of concerns that the insecticide was toxic to birds and because effective alternatives were available. However, it was registered by EPA in January, 2001 for use on non-food crops in greenhouses. In 2005, EPA established a tolerance for residues of chlorfenapyr in or on all food commodities. Chlorfenapyr works by disrupting the production of Adenosine triphosphate, specifically, "Oxidative removal of the N-ethoxymethyl group of chlorfenapyr by mixed function oxidases forms the compound CL 303268. CL 303268 uncouples oxidative phosphorylation at the mitochondria, resulting in disruption of production of ATP, cellular death, and ultimately organism mortality."
References
- US EPA (2001). "Pesticide Fact Sheet: Chlorfenapyr". http://www.epa.gov/opprd001/factsheets/chlorfenapyr.pdf. Retrieved 2007-04-18.
- US EPA (2000). "Decision Memorandum: Denial of Registration of Chlorfenapyr for Use on Cotton". http://www.epa.gov/opprd001/chlorfenapyr/chlorfenapyr.pdf. Retrieved 2006-07-28.
External links
Categories:- Insecticides
- Nitriles
- Pyrroles
- Organobromides
- Organochlorides
- Organofluorides
- Ethers
Wikimedia Foundation. 2010.
